The reaction is extremely exothermic, producing a bright yellow light and a great amount of heat energy. NaCl forms easily through the combination of its component elements sodium and chlorine. Each ion is 6-coordinate and has a local octahedral geometry. The cell looks the same whether you start with anions or cations on the corners. NaCl is best thought of as a face-centered cubic array of anions with an interpenetrating fcc cation lattice (or vice-versa). 
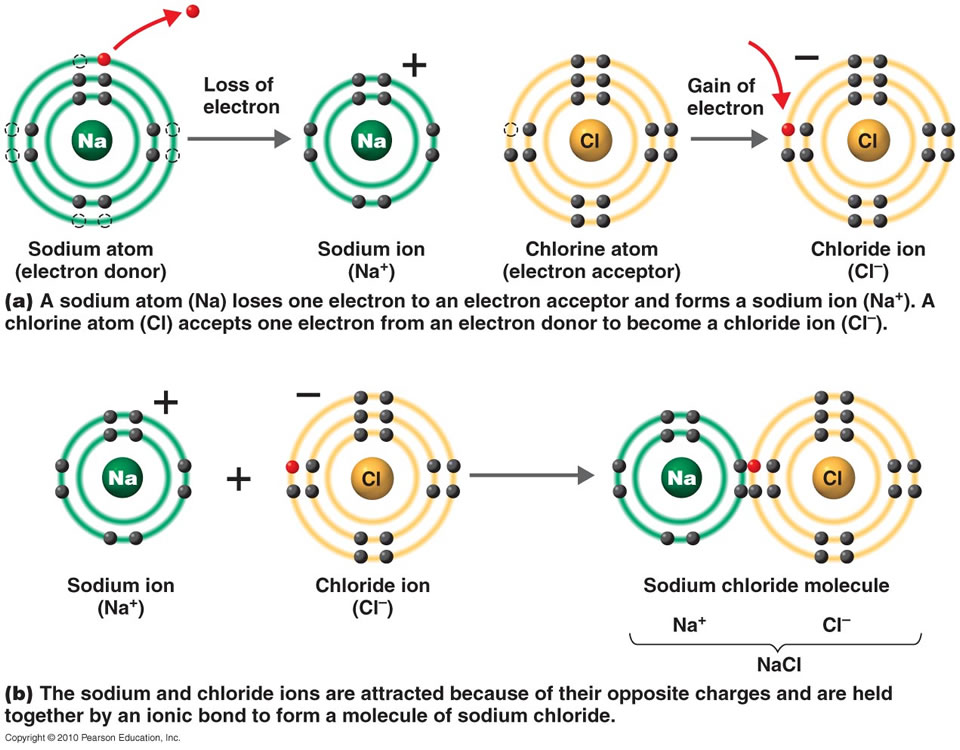
The chemical name of table salt is sodium chloride, NaCl. Perhaps the most common ionic compound people come across every day is table salt. (Right) In the crystal structure of sodium chloride, the purple spheres represent sodium cations, and the green spheres represent chloride anions Let’s discuss a few examples of ionic bonding in daily life. Moreover, Ionic compounds tend to form a crystal structure rather than being a molecule. Therefore, the term “ionic bond” is given when the ionic character is large enough to dominate over the polar covalent character. In fact, all ionic compounds have some degree of electron sharing (covalent nature). However, it is important to understand that electrons do not make a complete transfer from one atom or molecule to the other. The interspersing of positive and negative ions allows the ions to come close together until the repulsions balance the attractions. The two ions are then attracted to each other by electrostatic forces. The non-metal atom has a high electronegativity, and therefore, it readily gains electrons to form a negative ion or anion. When ionic bonds form, a metal donates one or more electrons, due to having a low electronegativity, to form a positive ion or cation. Ionic bonding tends to occur between electropositive metals and electronegative non-metals. It is one of the main types of chemical bonding along with covalent bonding and metallic bonding. The bond formed between oppositely charged ions, or two atoms due to a significant difference between their electronegativities, is known as an ionic bond. It is the measure of the tendency of an atom to attract the shared pair of electrons. One of such properties is electronegativity. The type of chemical bond formed depends upon the elements involved and their corresponding properties. Therefore, in simple terms, a chemical bond is known as a mutual attraction between two or more atoms that results in the redistribution of their outermost valance electrons. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. Hence, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. It is a well-established fact that opposite charges attract each other via a simple electromagnetic force. Chemical bonding is a key concept in chemistry to understand how these elements join together to form various compounds. Therefore they can conduct as their ions are free to move.All the matter present in the universe is made up of some combination of 118 elements we see on the periodic table. Ionic compounds dissolve in water easily, when they do this their lattice breaks up completely and water molecules surround the seperated ions. A cube of tightly packed alternate metal and non-metal ions. 
This is a regular repeating arrangement of metal and non-metal ions which creates compounds with very high melting points which conduct when molten or in solution but never when solid. Ionic compounds form what is known as a lattice structure. When these two charged particles come together they form an ionic bond because the positive magnesium ion is attracted to the negatively charged chloride ion. To become stable it must gain an electron to obtain a full outer energy level. Non-metals form negative ions because they gain electrons to become stable.Ĭhlorine (Cl) has an electron arrangement 2,8,7. They change into ions with a two positive charge. To become stable it must lose its two outer electrons to obtain a full outer energy level.Ītoms are neutral because they have equal numbers of protons and electrons however, when they lose two electrons they are no longer neutral. Magnesium (Mg) has the electron arrangement 2,8,2. Metals form positive ions because they lose electrons to become stable. The ionic bond is the electrostatic force of attraction between a positively charged metal ion and a negatively charged non-metal ion. Ionic bonds are formed between a metal and non-metal, for example sodium chloride.Īn atom of sodium will lose an electron and form a positive ion.Īn atom of chlorine will gain an electron and form a negative ion. 
Learn about the basic structure of an ion, related to atomic number and mass.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
